Nuclear OncologyNuclear Oncology is a newer field of medicine that combines targeted therapies based on a diagnostic test. Nuclear medicine uses targeted radiation to kill cancer cells from within. The diagnostic component determines and defines the type and extent of the disease which allows decisions to be made on timing, quantity, and choice of treatment options. The therapy component focuses on individualizing the treatment by targeting therapy to a patient’s specific disease using a therapeutic agent that attracts directly to the site without exposure to normal tissue. The treatment highly individualized and is very specific to the area it treats. |
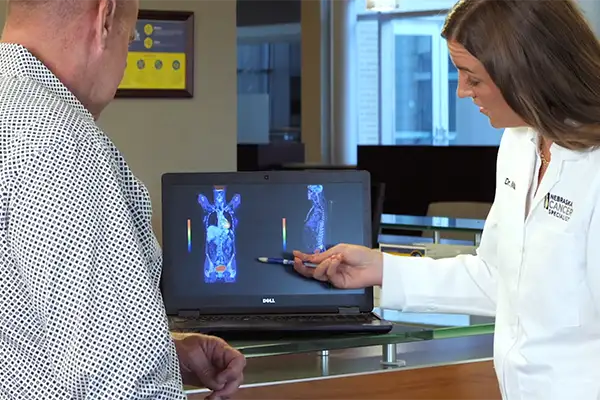
The nuclear oncology program at NCS is the first of its kind in the Midwest.
- Dedicated to providing one-on-one physician lead team to patient care
- Private outpatient setting with convenient drive up parking, nine treatment rooms
- PET/CT, SPECT/CT and Nuclear imaging along with two Radiopharmacy labs
- Mobile nuclear oncology team currently serving locations across the Midwest
-
What is Nuclear Oncology?
Watch an overview with NCS Nuclear Medicine Physician, Samuel Mehr MD.
Watch Video
Contact Us Now
To learn more or inquire about Nebraska Cancer Specialists Nuclear Oncology Center, give us a call at 402.334.4773.
Purpose • Innovation • Relationship • Excellence
© 2024 Nebraska Cancer Specialists • Privacy Policy • design by Chip Thompson
Meet DOT.